My info;
Data Zone
Classification: Zinc is a transition metal Color:bluish silver Atomic weight: 65.41
State:solid Melting point:419.53 oC, 692.68K Boiling point:910 oC, 1183K Electrons: 30 Protons: 30 Neutrons in most abundant isotope: 34 |
Discovery of Zinc
Zinc ores have been used to make brass (a mixture ofcopper and zinc) and other alloys since ancient times. A zinc alloy comprising 87.5% zinc was discovered in an ancient site in Transylvania. Zinc smelting began in the 12th century in India by reducing calamine (zinc carbonate, ZnCO3) with wool and other organic materials. The element name is reported to come from the old German word ‘zinke’ meaning pointed; a reference to the sharp pointed crystals formed after smelting. Credit for isolating the metal is usually given to Andreas Marggraf in 1746, in Berlin. He heated a mixture of calamine ore and carbon in a closed vessel without copper to produce the metal. |
Interesting Facts;
~Zinc is used to galvanize iron to inhibit corrosion.
~Zinc forms many alloys with other metals such as brass, German silver, nickel silver, typewriter metal and solders. ~Zinc alloys are used in die-castings for the car industry.
~The metal acts as both the container and as an electrode in zinc-carbon batteries. The drawback of this dual use is that the electrode is consumed when the battery is in use, getting thinner until eventually the battery starts leaking.
~Zinc is an essential trace element for animals and plants.
~Zinc oxide, a white powder, is a versatile compound that has many uses.
~It is used in sun block, make-up and in ointments such as calamine lotion.
~It is also used in the rubber industry, concrete manufacturing and in paints.
Harmful effects:
~Zinc is not considered to be particularly toxic.
~Zinc is essential for good health, but excess zinc is toxic and can cause nausea.
Characteristics:
~Zinc is a bluish-silver, lustrous metal that tarnishes in moist air, producing a layer of carbonate. It is somewhat brittle at room temperature but malleable above 100 oC.
~Zinc reacts with both acids and alkalis.
~It is a fair conductor of electricity, and burns in air with a bright bluish-green flame producing white clouds of the oxide.
~Zinc forms many alloys with other metals such as brass, German silver, nickel silver, typewriter metal and solders. ~Zinc alloys are used in die-castings for the car industry.
~The metal acts as both the container and as an electrode in zinc-carbon batteries. The drawback of this dual use is that the electrode is consumed when the battery is in use, getting thinner until eventually the battery starts leaking.
~Zinc is an essential trace element for animals and plants.
~Zinc oxide, a white powder, is a versatile compound that has many uses.
~It is used in sun block, make-up and in ointments such as calamine lotion.
~It is also used in the rubber industry, concrete manufacturing and in paints.
Harmful effects:
~Zinc is not considered to be particularly toxic.
~Zinc is essential for good health, but excess zinc is toxic and can cause nausea.
Characteristics:
~Zinc is a bluish-silver, lustrous metal that tarnishes in moist air, producing a layer of carbonate. It is somewhat brittle at room temperature but malleable above 100 oC.
~Zinc reacts with both acids and alkalis.
~It is a fair conductor of electricity, and burns in air with a bright bluish-green flame producing white clouds of the oxide.
Family Members;
Group; 12 2B
Family members; 3, Cadmium, mercury and ununquadium.
Family Properties; Zinc family is reactive, The family is made up of metals, and the family is intert.
Status;
The abundance of zinc in the Earth's crust is estimated to be about 0.02 percent. That places the element
about number 24 on the list of the elements in terms of their abundance. A process for extracting zinc from its ores was apparently invented in India by the 13th century. Zinc never occurs as a free element in the earth. Some of its most important ores are smithsonite, or zinc spar or zinc carbonate (ZnCO 3 ); sphalerite, or zinc blende or zinc sulfide (ZnS); zincite, or zinc oxide (ZnO); willemite, or zinc silicate (ZnSiO 3 ); and franklinite [(Zn,Mn,Fe)O
(Fe,Mn 2 )O 3 ]. The largest producer of zinc ore in the world today is Canada. Other important producing nations are Australia, China, Peru, the United States, and Mexico. In the United States, more than half of the zinc produced comes from Alaska. Other important producing states are Tennessee, Missouri, Montana, and New York.
Physical properties
Zinc is a bluish-white metal with a shiny surface. It is neither ductile nor malleable at room temperature. Ductile means capable of being drawn into thin wires. Malleable means capable of being hammered into thin sheets. At temperatures above 100°C (212°F), however, zinc becomes somewhat malleable.
Zinc's melting point is 419.5°C (787.1°F) and its boiling point is 908°C (1,670°F). Its density is 7.14 grams per cubic centimeter. Zinc is a fairly soft metal. Its hardness is 2.5 on the Mohs scale. The Mohs scale is a way of expressing the hardness of a material. It runs from 0 (for talc) to 10 (for diamond).
Chemical properties
Zinc is a fairly active element. It dissolves in both acids and alkalis. An alkali is a chemical with properties opposite those of an acid. Sodium hydroxide ("common lye") and limewater are examples of alkalis. Zinc does not react with oxygen in dry air. In moist air, however, it reacts to form zinc carbonate. The zinc carbonate forms a thin white crust on the surface which prevents further reaction. Zinc burns in air with a bluish flame.
Family members; 3, Cadmium, mercury and ununquadium.
Family Properties; Zinc family is reactive, The family is made up of metals, and the family is intert.
Status;
The abundance of zinc in the Earth's crust is estimated to be about 0.02 percent. That places the element
about number 24 on the list of the elements in terms of their abundance. A process for extracting zinc from its ores was apparently invented in India by the 13th century. Zinc never occurs as a free element in the earth. Some of its most important ores are smithsonite, or zinc spar or zinc carbonate (ZnCO 3 ); sphalerite, or zinc blende or zinc sulfide (ZnS); zincite, or zinc oxide (ZnO); willemite, or zinc silicate (ZnSiO 3 ); and franklinite [(Zn,Mn,Fe)O
(Fe,Mn 2 )O 3 ]. The largest producer of zinc ore in the world today is Canada. Other important producing nations are Australia, China, Peru, the United States, and Mexico. In the United States, more than half of the zinc produced comes from Alaska. Other important producing states are Tennessee, Missouri, Montana, and New York.
Physical properties
Zinc is a bluish-white metal with a shiny surface. It is neither ductile nor malleable at room temperature. Ductile means capable of being drawn into thin wires. Malleable means capable of being hammered into thin sheets. At temperatures above 100°C (212°F), however, zinc becomes somewhat malleable.
Zinc's melting point is 419.5°C (787.1°F) and its boiling point is 908°C (1,670°F). Its density is 7.14 grams per cubic centimeter. Zinc is a fairly soft metal. Its hardness is 2.5 on the Mohs scale. The Mohs scale is a way of expressing the hardness of a material. It runs from 0 (for talc) to 10 (for diamond).
Chemical properties
Zinc is a fairly active element. It dissolves in both acids and alkalis. An alkali is a chemical with properties opposite those of an acid. Sodium hydroxide ("common lye") and limewater are examples of alkalis. Zinc does not react with oxygen in dry air. In moist air, however, it reacts to form zinc carbonate. The zinc carbonate forms a thin white crust on the surface which prevents further reaction. Zinc burns in air with a bluish flame.
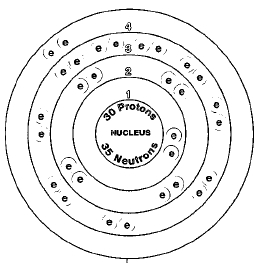
Number of electrons in each shell of bohr model;
Shell 1, two electrons.
Shell 2, eight electrons.
Shell 3, eighteen electrons.
Shell 4, two electrons.
Valence Electrons: 4s2
Zinc want to lose electrons when bonding. It loses
Oxidation States: 2
Zinc can bond with sulfur, oxygen and chlorine. Zinc is a transition metal
Compounds form and formulas of the compound;
A number of zinc compounds have important uses. Some examples are the following:
~Zinc deficiency can interfere with a plant's ability to reproduce.
~Zinc acetate (Zn(C 2 H 3 O 2 ) 2 ): wood preservative; dye for textiles; additive for animal feed; glazing for ceramics
~Zinc arsenate (Zn 3 (AsO 4 ) 2 ): wood preservative; insecticide
~Zinc borate (ZnB 4 O 7 ): fireproofing of textiles; prevents the growth of fungus and mildew
~Zinc chloride (ZnCl 2 ): solder (for welding metals); fireproofing; food preservative; additive in antiseptics and deodorants; treatment of textiles; adhesives; dental cement; petroleum refining; and embalming and taxidermy products.
~Zinc fluorosilicate (ZnSiF 6 ): mothproofing agent; hardener for concrete
~Zinc hydrosulfite (ZnS 2 O 4 ): bleaching agent for textiles, straw, vegetable oils, and other products;
~Zinc alloys are used in the production of electrical fuses.
brightening agent for paper and beet and cane sugar juice
zinc oxide (ZnO): used in rubber production; white pigment in paint; prevents growth of molds on paints; manufacturer of glass; photocopy machines; production of many kinds of glass, ceramics, tile, and plastics
~Zinc phosphide (Zn 3 P 2 ): rodenticide (rat killer)
~Zinc sulfate (ZnSO 4 ): manufacture of rayon; supplement in animal feeds; dyeing of textiles; and wood preservative
Shell 1, two electrons.
Shell 2, eight electrons.
Shell 3, eighteen electrons.
Shell 4, two electrons.
Valence Electrons: 4s2
Zinc want to lose electrons when bonding. It loses
Oxidation States: 2
Zinc can bond with sulfur, oxygen and chlorine. Zinc is a transition metal
Compounds form and formulas of the compound;
A number of zinc compounds have important uses. Some examples are the following:
~Zinc deficiency can interfere with a plant's ability to reproduce.
~Zinc acetate (Zn(C 2 H 3 O 2 ) 2 ): wood preservative; dye for textiles; additive for animal feed; glazing for ceramics
~Zinc arsenate (Zn 3 (AsO 4 ) 2 ): wood preservative; insecticide
~Zinc borate (ZnB 4 O 7 ): fireproofing of textiles; prevents the growth of fungus and mildew
~Zinc chloride (ZnCl 2 ): solder (for welding metals); fireproofing; food preservative; additive in antiseptics and deodorants; treatment of textiles; adhesives; dental cement; petroleum refining; and embalming and taxidermy products.
~Zinc fluorosilicate (ZnSiF 6 ): mothproofing agent; hardener for concrete
~Zinc hydrosulfite (ZnS 2 O 4 ): bleaching agent for textiles, straw, vegetable oils, and other products;
~Zinc alloys are used in the production of electrical fuses.
brightening agent for paper and beet and cane sugar juice
zinc oxide (ZnO): used in rubber production; white pigment in paint; prevents growth of molds on paints; manufacturer of glass; photocopy machines; production of many kinds of glass, ceramics, tile, and plastics
~Zinc phosphide (Zn 3 P 2 ): rodenticide (rat killer)
~Zinc sulfate (ZnSO 4 ): manufacture of rayon; supplement in animal feeds; dyeing of textiles; and wood preservative